Study Goal
Identify and validate intraoperative TTFM & HFUS findings (e.g. threshold values of mean graft flow, maximum flow capacity, flow curve pattern, morphological issues) that are related to problems in: graft inflow, graft itself or graft outflow that
- are predictive of future graft events (loss of patency)
- may be a reason for corrections during the index surgery
- are obtained with safe and effective techniques
A health economic analysis will also be performed.
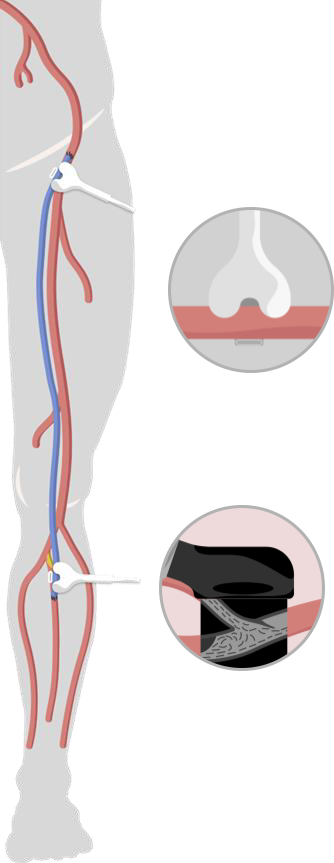
Protocol Overview
- Open, prospective multi-center cohort study to address the intra-operative benefits and prognostic value of TTFM & HFUS in open bypass in CLTI patients
- Standardized TTFM & HFUS assessments
- Surgical procedures according to routine practice
- Graft patency follow-up using Duplex ultrasound at 1, 3, 6 and 12 months
Patient Population
CLTI patients scheduled to undergo a vein bypass with or without arterial inflow reconstruction to a below-knee and above ankle target (including popliteal with single vessel arterial run-off)
The Medistim MiraQ system is the platform for the TTFM and HFUS measurements. A standardized protocol for completion TTFM includes the intragraft delivery of the vasodilator papaverine. The trial will test specific threshold values for prediction of 1 year patency.






